EksoNR -
Robotic Therapy, Simplified
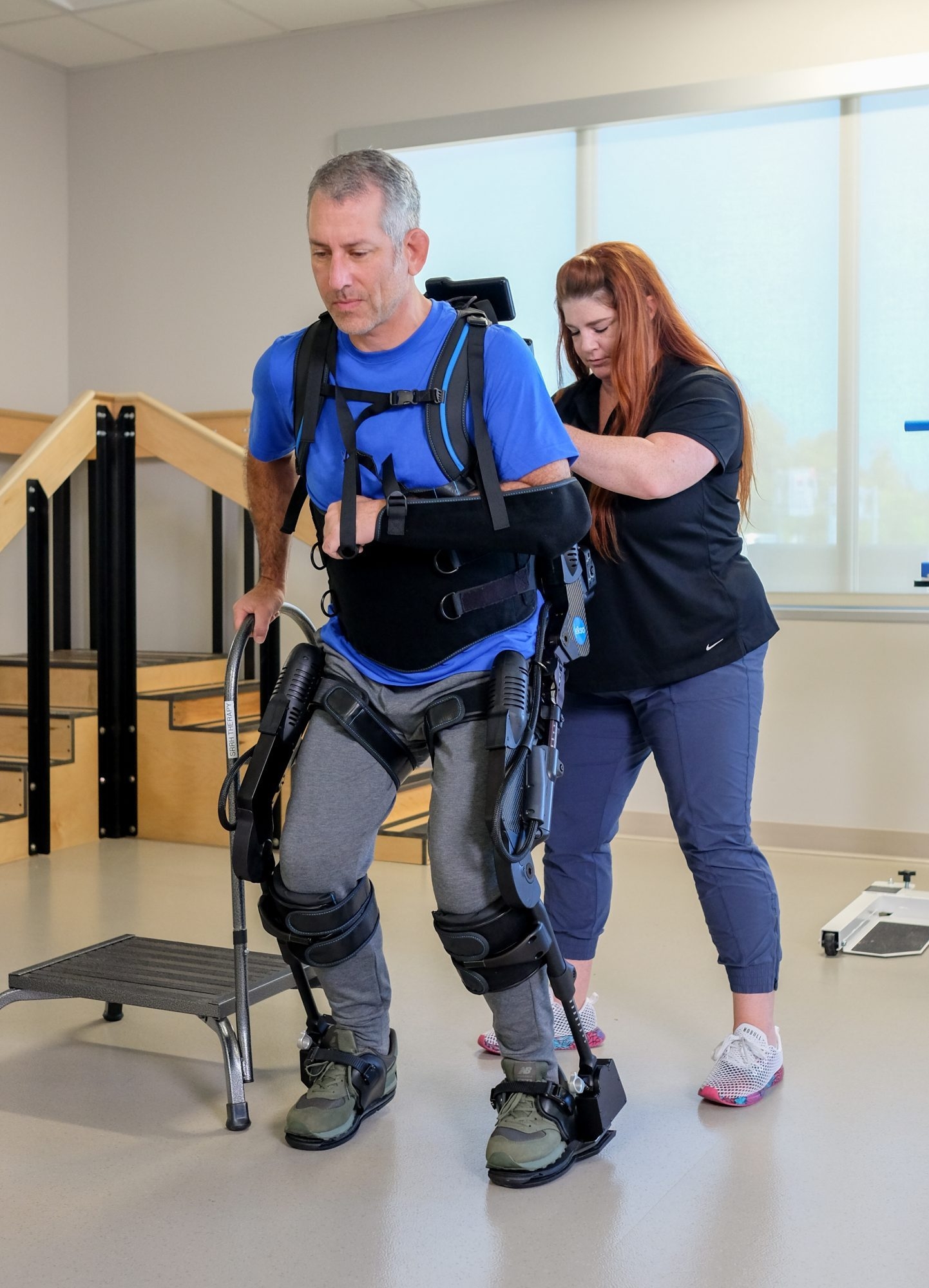
EksoNR, our robotic exoskeleton designed for rehabilitation settings, enables high-acuity patients to stand and walk within minutes – delivering the intensity, repetition, and early mobility needed to drive neuroplasticity and faster recovery*. With the new intelligent, real-time gait feedback and a streamlined setup, EksoNR helps therapists maximize every session while reducing physical strain. The result: more efficient therapy, accelerated functional outcomes, and a safer, more empowering experience for patients when it matters most.
EksoNR + GaitCoach
EksoNR’s GaitCoach software makes our robotic exoskeleton even more intuitive, easier to use, and faster to set up and train while maintaining broad customization for each patient. EksoNR with GaitCoach provides insight to simplify complex therapy – giving therapists more confidence, clarity, and control with every step. Therapists can begin using EksoNR one-on-one with patients immediately after training, improving efficiency for patients of all functional levels.
Key Benefits:
- See what matters through new real-time deviation detection
- Determine where to focus with clear, actionable guidance
- Work more confidently with automatic insights
- Understand progress instantly with in-session feedback
- Stay fully in control – GaitCoach enhances, not replaces, your own clinical expertise on how to best treat patients
Designed to help neuro rehabilitation teams do more
Start Gait Training Sooner
Mobilize high-acuity patients quickly and safely, enabling standing and walking earlier in the recovery process. Early mobilization supports better outcomes and improved patient confidence.
Deliver The Right Intensity For Recovery
Achieve hundreds to thousands of steps per session, providing the dosage required to drive neuroplasticity and meaningful functional gains.
Enhance Clinical Decision-Making
EksoNR’s new GaitCoach software delivers real-time feedback on gait and balance, along with immediate feedback that helps therapists adjust and progress treatment with precision.
Maximize Therapist Efficiency
Providing extra support and oversight, EksoNR reduces the physical demands of gait training and simplifies advanced rehab technology – allowing clinicians to focus on patient care and rehab potential.
Clinically validated performance
As the first exoskeleton FDA-cleared for stroke, acquired brain injury, MS and SCI and backed by more than 200 published articles, Ekso has demonstrated safety and improved rehabilitation outcomes – giving clinicians and administrators confidence in both adoption and results. *
For inpatient facilities
Standardize high-quality therapy across clinicians, improve session efficiency, and help patients progress faster – supporting shorter inpatient lengths of stay and differentiated rehab programs
For outpatient facilities
EksoNR is utilized as an advanced therapeutic tool to enhance endurance and increase walking distance, ultimately refining functional ambulation. It’s user-friendly features support continued patient progression, helping clinicians maximize rehabilitation outcomes while promoting a seamless continuum of care from hospital to home.
Create a better patient experience
Patients feel safe, supported, and motivated – often achieving more, earlier in their recovery than they thought possible after a life-altering event.
Backed by data from over 40
industry-leading research partners
EksoNR was developed by clinicians for clinicians. With more than 2,000 patients participating in over 185 clinical trials, EksoNR is the most researched mobile exoskeleton on the market. Learn how training with EksoNR can improve the limitations in walking ability caused by neurological disorders, along with their physiological and psychological consequences, thus improving patients' quality of life.
See our evidence
Features
GAITCOACH SOFTWARE
GaitCoach analyzes components of walking and notifies the therapist what to focus on to improve gait quality.
CLINICIAN CONTROL
Modify swing and stance assistance levels in real time based on patient goals and GaitCoach provided feedback.
POSTURE SUPPORT
EksoNR helps patients bear only their own weight while assisting with postural alignment through the trunk so you can treat patients through the continuum of care.
DATA CAPTURE
View session-specific data including walking time, distance, and speed, securely saved to a cloud-based dashboard for easy analytics.
PATIENT ENGAGEMENT
Patients actively participate to correct gait deviations based on visual and auditory feedback provided by GaitCoach.
PROGRESSIVE GAIT TRAINING
Intelligent software continuously monitors the walking pattern and guides the therapist to progress the patient to maximize intensity.
MORE THAN WALKING
EksoNR includes pregait activities to help patients balance, weight shift, squat, and step in place before walking.
EksoNR FAQs
EksoNR is the only exoskeleton designed specifically by clinicians for clinicians and features software incorporating early mobility tools and independent walking support. EksoNR is FDA cleared for stroke, acquired brain injury, MS, and spinal cord injury. It is the only exoskeleton with ABI and MS clearance. For full indications for use, please visit here: link
EksoNR fits patients under 220 pounds and between 5′ and 6’4″.
EksoNR weighs approximately 55 lbs with batteries. The device is self-weight bearing and carries its own weight. The patient is bearing full weight while in the device but is not responsible for carrying the weight of EksoNR. It is also portable and easily transported in the clinic using a cart or wheelchair.
No, EksoNR is only available in certified rehabilitation centers. Find a center near you with our interactive map. If you are interested in a personal exoskeleton for home and community use, check out our Ekso Indego Personal for more information.
EksoNR is the only exoskeleton designed specifically by clinicians for clinicians and features software incorporating early mobility tools and independent walking support. EksoNR is meant to be used across the continuum of care in inpatient and outpatient rehab. The ideal goal with EksoNR is to have the patient “walk out” of the device and back into their community, if possible. In addition, Ekso Bionics provides clinical, technical, training, and marketing support to ensure your Ekso program is as successful as possible.
Ekso Bionics exoskeletons are only available for patients from 5′ to 6′ 4″ and is FDA cleared for use for those who are 18 years old and over. Please contact Customer Relations for more information.