A stroke is a medical emergency, capable of damaging the brain through interruption of its blood supply. There are many symptoms of stroke including difficulties walking, speaking, and understanding, as well as numbness of the face, arm, or leg. When it comes to treating subacute stroke in an inpatient rehabilitation setting, Dr. Crissy Voigtmann (Doctor of Physical Therapy and board-certified Neurological Clinical Specialist), Dr. CJ Curran (Doctor of Physical Therapy, certified brain injury specialist and board-certified Neurological Clinical Specialist), and Jose Dominguez (Director of Rehabilitation, Orlando Health) give professional insight on EksoNR, the first FDA-cleared exoskeleton for stroke and spinal cord injury, and the only FDA-cleared exoskeleton for acquired brain injury.
Orlando Health ORMC Institute for Advanced Rehabilitation (IAR) is a CARF accredited inpatient and outpatient rehab unit partnered with Ekso Bionics since 2019. Currently, they have four Level-Two trained therapists at the inpatient rehabilitation, and two Level-Two trained therapists at their outpatient rehabilitation.
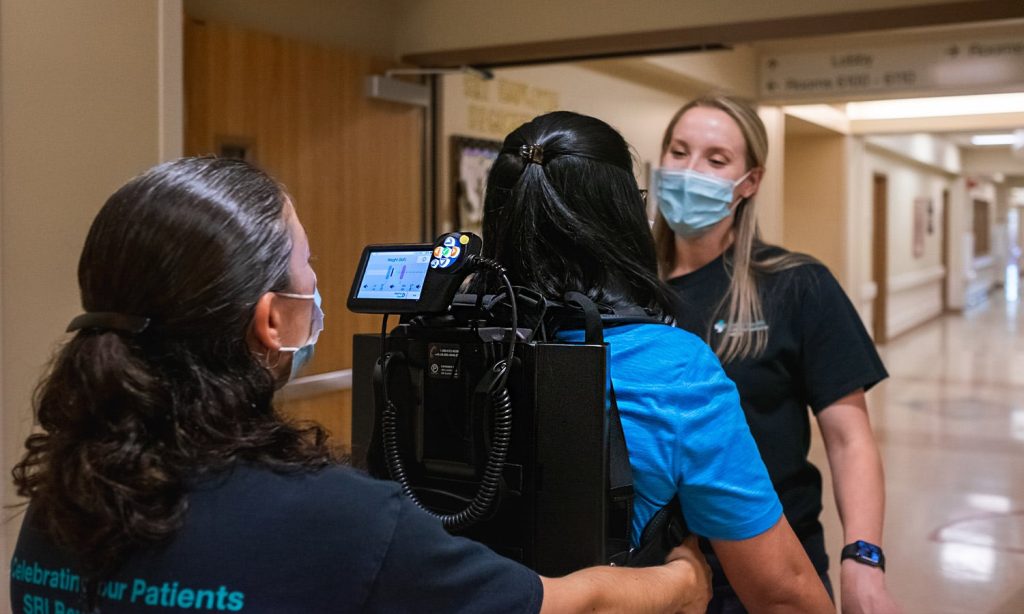
What is EksoNR?
Currently, EksoNR is the only exoskeleton that is FDA-cleared for acquired brain injury. It is the first FDA-cleared exoskeleton and extremely adaptive with short-term implications as well as long-term ones. The device is capable of progressing through programming as a stroke patient progresses in recovery. This gives the long-term capability to adapt from session to session, not only for patients with strokes but spinal cord injuries or TBI.
Who can benefit from EksoNR?
In 2020, IFR had 310 stroke patients arrive at the unit. On average, they had a 1.58 acuity level, based on a case-mix index equation that determines the acuity level for inpatient rehab. Typically a stroke patient stayed at the unit for 15.7 days. In order to determine mobility scores upon admission, Crissy and CJ used Care (formerly known as FIM), which is a standardized test that all inpatient rehabilitation facilities have to use for all diagnoses.
Additional Care scores from the facility include:
- Average Care Score: 2.9 (Max Assist)
- Average Walking Score (per 50ft): 2.2 (Max Assist)
- Average Walking Score (per 150ft): 1.7 (Total Assist)
- Average Stair Score: 1.2 (Total Assist)
At IAR, these scores were what the average numbers looked like for all stroke patients and are important to know to where those who used Ekso started from at baseline.
[The following is condensed from the full webinar of Dr. Crissy Voigtmann and Dr. CJ Curran.]
What were the deciding factors in having Ekso a part of the program at Orlando Health?
As Dr. Crissy Voigtmann and Dr. CJ Curran were in the decision-making process of having Ekso become a part of their program, they were asking themselves: “Are we doing enough? Are we providing these patients with enough repetitions at a high enough intensity level to elicit the neural change that needs to happen during this short length of stay that we have?”
In general, the answer to the questions they asked was no. They ultimately decided there was not an exact dosage that has been established for intensity or for repetition, but there was a growing body of evidence that demonstrates for gait in particular; that stepping practice needs to be in the thousands a day for optimal improvement.
Observational studies have shown that in inpatient rehab facilities (places that have stroke programs), stroke patients are taking an average of 250 steps a day. To further reinforce that point, these same studies have shown that stroke patients on average spend greater than 50% of the day in bed and greater than 60% of their time alone.
Not only were they not utilizing the active time with these folks to provide high-intensity therapy that is necessary for neural training, but even during the passive time, they realized they were not providing the necessary environment, whether it be with engagement or stimulation that is necessary for improvement as well.
They determined the best solution was integrating Ekso into their program. This was the bridge. This bridged the gap between that dilemma and that solution. For them, Ekso allowed them to accomplish those goals on day one and early, especially for their folks who are significantly impaired, the ones with a lot of deficits. These were folks, in their experience, that are really difficult to gait train. These are the people that unfortunately tend to fall through the cracks and would get low level therapy or low intensity therapy otherwise.
How has Ekso changed training for the patients?
Because of Ekso, they were able to get individuals up early and on their feet for extended periods of time, participating in high-intensity therapy from the very beginning. There are certain positions that would take two or three therapists, or three pairs of hands, to get a patient to accomplish. But one therapist is able to do it whilst focusing on other deficits as well, thanks to Ekso.
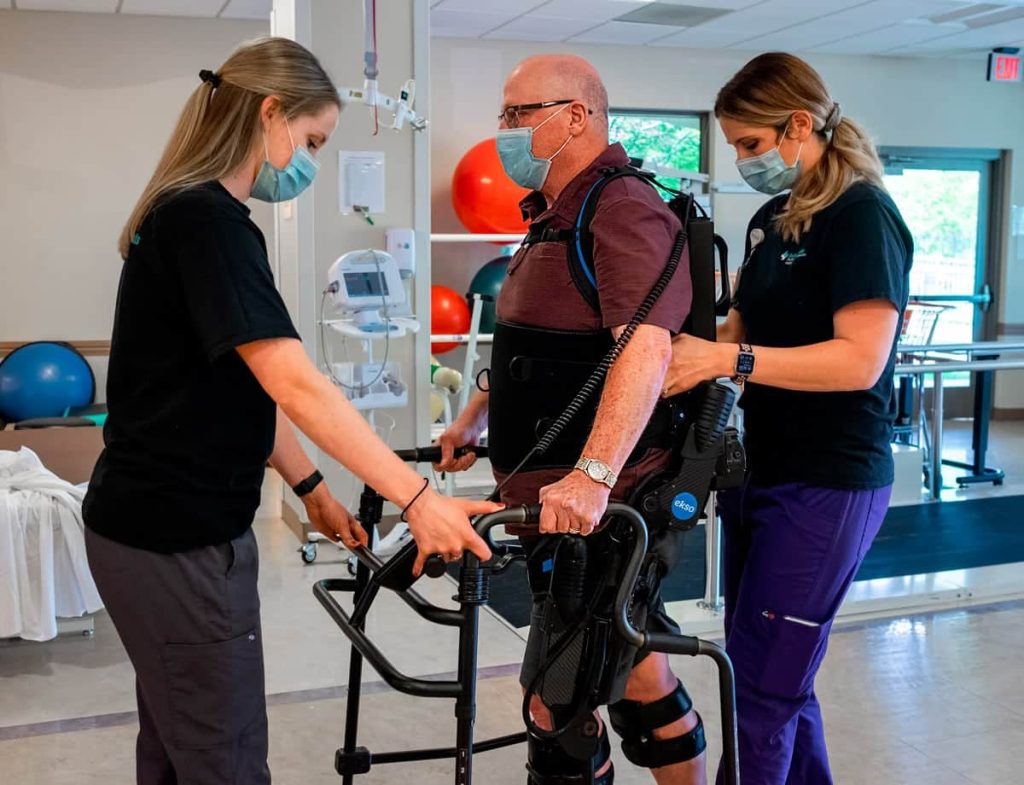
Who are the candidates for Ekso?
The stroke patients primarily receiving early Ekso intervention:
- Dense Hemiparesis: 0-⅖ MMT throughout affected lower extremity
- Pusher Syndrome
- Significant Postural Control and Awareness deficits
- Motor planning and Sequencing deficits
Other patient specific factors leading to early Ekso utilization:
- Obesity/Height (under 220lbs, under 6’5” approximately)
- Impulsivity
- Cognitive deficits
The overall message is these patients are individuals who are very difficult to manually facilitate. They are very difficult to provide the manual techniques that are necessary to hit high repetitions or to hit high intensity levels. As a result, they are going into the exoskeleton.
Stay Tuned For More Blogs About EksoNR or Click Here to View The Webinar in Full